Copper Oxide Reduction Technology

Copper Oxide Reduction to Elemental Copper
- Pre-cleaning a conductive interconnect structure is critical in the integrated circuits (ICs).
- The copper conductive layer must be cleaned before subsequent deposition.
- Spruce Science present the patent pending solution for copper oxide reduction using gas-phase ethanol.
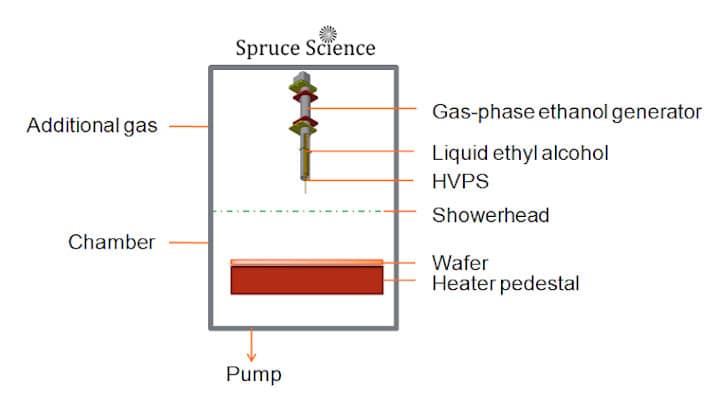
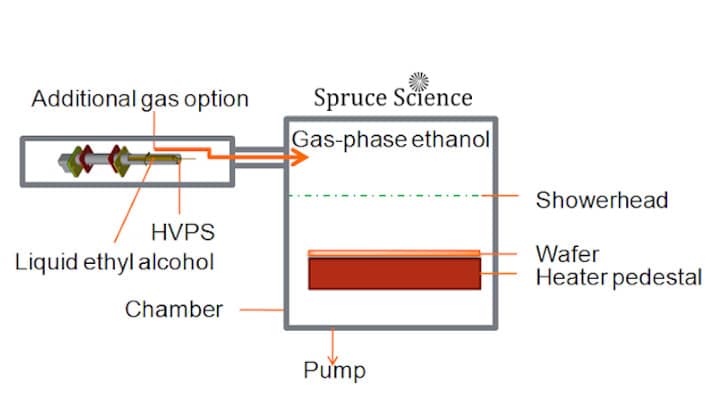
- Gas-phase ethanol is generated by applying high voltage to liquid ethyl alcohol (C2H5OH).
- The value in this preclean technique is that it will prevent damage to low-k dielectrics.
- Using high voltage to generate gas-phase ethanol will provide precisely controlled delivery of gas-phase ethanol for copper oxide reduction.
- High purity vapor-phase ethanol is generated and will prevent unwanted contaminations.
Table of Contents
Background
As a conductor, copper has many desirable material properties and that makes it attractive in the semiconductor fabrication process and beyond. Using copper as an interconnect in integrated circuits (ICs) can improve performance. Some of the benefits of using copper in thin-film applications are the low bulk electrical resistivity of 1.7 µOhm, high melting point at 1083 °C, and electromigration resistance.
Despite the benefits, there are challenges associated with copper as an interconnect in integrated circuits (ICs). One of the concern is that copper has high diffusivity in silicon with a coefficient of 2.0×10-5 cm2s-1 at 500° C. The other concern is that copper forms an oxide but it does not form a self-limiting oxide layer that is stable. The oxide growth mechanism is that cuprous oxide, Cu2O, forms with the metallic copper. Then cupric oxide (CuO) nucleates on top of that Cu2O layer and continues to grow under certain conditions.
Such characteristic of copper oxide layer formation and unstable oxide growth result in poor adhesion to barrier films, poor electromigration behavior, and poor via resistance between metal contacts that negatively affects IC performance. Therefore, the oxide layer is undesirable and degrades copper interconnect performance.
For successful usage of copper as a thin-film in the semiconductor fabrication process, copper oxide reduction is absolutely required and the oxide layer needs to be removed before subsequent depositions. One method of copper oxide reduction is to use hydrogen. The reduction of copper oxide to elemental copper using hydrogen plasma is effective and efficient. However, the problem with this cleaning process is that it can cause damage to low-k polymer dielectric materials. For the type of applications where low-k dielectrics are exposed, there will be compatibility issues with using hydrogen plasma. In such a scenario, damage to low-k dielectrics occurs during the in-situ cleaning process because of hydrogen radicals.
DESCRIPTION OF USING HIGH VOLTAGE TO GENERATE GAS-PHASE ETHANOL FOR COPPER OXIDE REDUCTION WITHOUT CAUSING DAMAGE TO LOW-K DIELECTRICS
The invention from Spruce Science provide a method for copper oxide reduction to elemental copper without causing damage to low-k dielectrics. This method uses high voltage to generate gas-phase ethanol from liquid ethyl alcohol (C2H5OH). The ethanol vapor is delivered to the process chamber where the wafer is heated above 150°C. This method of precleaning the copper conductive layer is required before subsequent depositions and will prevent damage to low-k dielectrics. This capability is important for the semiconductor and other thin-film fabrication process where feature sizes and performance are being optimized for integrated circuits.
The benefit of this preclean technique is that it allows for copper oxide reduction without damage to low-k dielectric. It allows for precleaning a conductive interconnect structure in the integrated circuits (ICs).
One conventional method to generate gas-phase ethanol would be from bubbling a carrier gas. Another method of gas-phase ethanol generation relies on the fact that ethanol has high vapor pressure and that characteristic can be utilized to deliver ethanol gas at lower pressure. However, this delivery technique is passive and slow since the flow rate is limited by the vapor pressure. Above all, it offers limited control over the process.
With our technique, gas-phase ethanol is generated from liquid ethanol by applying high voltage directly to the liquid. The presence of the high electric field create a characteristic Taylor Cone formation. From there, liquid ethanol breaks up into droplets under the presence of an intense electric field. The liquid droplets continue to get smaller until the droplets eventually become vapor-phase ethanol. The gas-phase ethanol is then delivered to the wafer substrate that is heated above 150 °C for effective cleaning and copper oxide reduction.
Spruce Science method of using high voltage to generate gas-phase ethanol will provide more controlled delivery of gas-phase ethanol, which is important in semiconductor manufacturing process.
Using high voltage to generate gas-phase ethanol is more effective at higher pressures and that will improve the cleaning process.
High purity vapor-phase ethanol will be generated from liquid ethanol. This will prevent unwanted contaminations, which is a top priority in semiconductor processing.