Electrospray Ionization using a Precision High Voltage Power Supply for Optical Emission Spectroscopy
Demonstration Notes
Introduction
There are many fascinating high voltage applications in the modern society. One of the more interesting applications is in analytical instruments such as mass spectrometers where high voltage power supplies are integrated in a variety of ways. Precision high voltage power supply can be used to create ions, accelerate ions, form and shape the ion beams, and power special detectors with high gains. There are varying types of mass spectrometers and actual configuration depend on the application.
One unique application in particular that will be the focus of this article is to use the precision high voltage power supply for electrospray ionization at atmospheric pressure. The electrospray ionization was first reported in an academic article by Masamichi Yamashita and John Fenn in 1984. Years later in 2002, John Fenn was awarded the Nobel Prize in Chemistry for the development of electrospray ionization technique for mass analysis of biological macromolecules.
Precision high voltage power supply from Spruce Science is used in this demonstration for electrospray ionization and high voltage is applied directly to sodium liquid solution to generate tiny sodium droplets. These charged sodium ions are then brought into a gas flame so that bright yellow light emission can be observed. The electrospray ionization coupled with optical emission spectroscopy is an analysis method that can be used to detect Alkali metals.
Spruce Science design and manufacture state-of-the-art precision high voltage power supply in Sunnyvale, California. The goal of this experiment is to demonstrate the usefulness of the highly integrated precision high voltage power supply that is compact, lightweight and vastly more versatile.


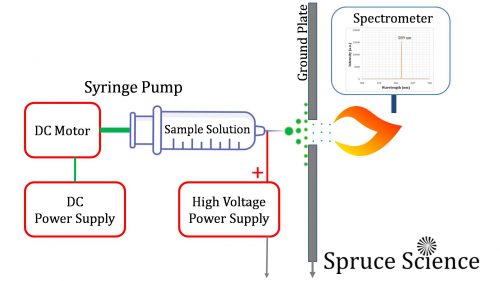
Schematic Illustration
The schematic illustration for the demonstration of electrospray ionization for optical emission spectroscopy is shown below and it is a two step process.
The left side of the ground plate is where electrospray ionization takes place. The sample solution is loaded inside a syringe pump and a DC motor slowly pushes the liquid through the needle. Using the precision high voltage power supply from Spruce Science, high voltage in the range of 2kV to 10kV is applied to the liquid sample directly for electrospray ionization to make dry and tiny droplets that are electrically charged.
The second part of the analysis is optical emission spectroscopy, which is illustrated on the right side of the ground plate. What happens here is that the gas-phase ions are brought into the gas flame and the excitation causes light emission with colors unique to the sample element. The spectral lines produced by the flames are then measured for analysis.
Power Supplies for Electrospray Ionization
Precision high voltage power supply used in this experiment is shown on the left side of the picture and is capable of producing 30kV at 1mA.
The compact form factor is great for usage in space-constrained systems since even the simplest experiments end up occupying a lot of space. It is also very convenient that the precision high voltage power supply is highly integrated because it simplifies the setup process. The only thing required from the user to get started is to plug-in.
On the right side of the picture is the DC power supply to energize the DC motor that drives the syringe pump.
Taylor Cone Formation during Electrospray Ionization
A lot of fascinating science can be observed during electrospray ionization if the tip of the needle is inspected under high magnification. When high voltage is applied from the needle, which is carrying a liquid flow, to the ground plate, there is this characteristic formation known as a Taylor cone. That Taylor cone is then pulled into a jet. From there, the jet is further disrupted into a series of tiny droplets. This characteristic formation is established when high voltage is balanced with pumping rate.

The general observation for the electrospray ionization process is if the applied voltage is too high and field extraction exceeds pumping rate, spraying jet tends to be off axis or causes arcing. However, if the applied electric field is too low, it leads to instability. For performance tuning, having an adjustable precision high voltage power supply is essential. The sign of the voltage drop across the liquid sample to the ground plate will determine which ion types we are looking at. In this demonstration, positive high voltage output is connected to the needle and the ground plate is negatively charged, meaning that positively charged ions are selected in the electrospray ionization process.
Flame Color Changes Immediately when Electric Field is Applied
When the syringe pump is running without the application of high voltage, droplets form then drips continuously from the tip of the needle. The sample used in this demonstration is sodium dissolved in water and isopropyl alcohol solution. The droplets forming at the tip of the needle are shown below.
It become interesting when positive 6kV is applied to the liquid sample. When high voltage power supply is enabled, electrospray ionization starts and sample ions are immediately delivered to the flame and color changes instantly. Flame color changes because thermal energy excites the sample molecules to a higher energy state and their transition back to a lower energy state leads to photon emission with unique wavelengths that are characteristic of the sample molecule. In this case, the flame produces a bright yellow color because of the sodium sample.


Slightly different view of the flame is shown below for observation. Sodium electrospray ionization coupled with a flame sure produced spectacular colors.


Mapping the Voltage Response for Electrospray Ionization
The gradual response will be presented by ramping the voltage from 0 to +6kV using the dial indicator on the desktop high voltage power supply. The sequence of images are displayed below with voltage output displayed above the needle.
When there is no voltage applied, droplets form, then drip straight down. As voltage increases, the effects of the electric field shows, and the shape of the droplets begin to change. When voltage is further increased to 3kV, droplets begin to launch towards the bottom side of the ground plate. The droplets continue to climb upward with increasing voltage. Mode of operation changes to spraying around 3.4 kV. At 4 kV, there are changes in flame color. The flame flickers between yellow and blue flame color so there are a lot of instability. By the time it reaches 5kV, the prominent bright yellow color emission resulting from sodium is established, clearly indicating that electrospray ionization is functional.








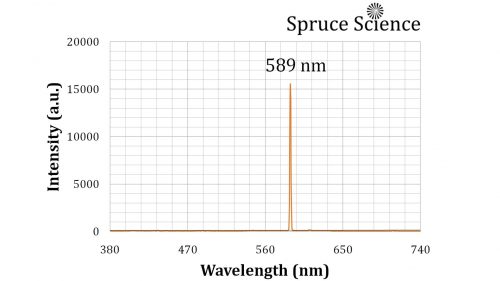
Optical Emission Spectroscopy
By using a spectrometer, it is possible to figure out the exact wavelength of the flame. The spectrum that was measured and displayed below shows light intensity on the y-axis and wavelength on the x-axis. The peak emission is at 589 nanometers and that is the bright yellow light that can be observed. The typical response of a human’s eye ranges from 380 nm to 740 nm wavelength on the electromagnetic spectrum.
This analysis method is such a great way to detect Alkali metals and demonstrate how useful it is to have a precision high voltage power supply that is compact, lightweight and versatile.
Conclusion
- Precision high voltage power supply can be used to apply electric fields to sample liquid solution to generate tiny droplets that are charged.
- When high voltage is applied from the needle, which is carrying a liquid flow, to the ground plate, there is this characteristic formation known as a Taylor cone. That Taylor cone is then pulled into a jet. From there, the jet is further disrupted into a series of tiny droplets.
- When aerosol generated by the electrospray ion source is brought to a gas flame, it produces light emission with unique colors.
- By using a spectrometer, the emission characteristics can be measured and analyzed.
- Sodium sample used in this demonstration creates a bright yellow light emission with peak emission at 589 nm.
- It is very beneficial and useful to have a reliable precision high voltage power supply that is adjustable from 0 to 30kV with 1mA output capability for experimentation.
Video Demonstration




