Paper Spray Ionization
Soft Ionization Technique for Mass Spectrometry
Demonstration Notes
Paper spray ionization technique introduction
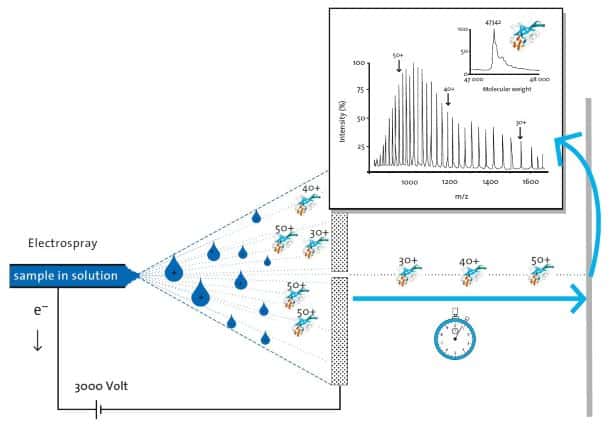
Paper spray ionization is a type of ion source that generate ions. This simple and effective ion source uses paper as the primary facilitator that deliver the sample in a controlled manner that produce ions under the influence of an intense electric field. The sample is applied to a piece of paper and solvent is added. It works best when the piece of paper has a sharp tip. When high voltage is applied to the paper that contain the solution, a microscopic jet emerges from the tip of the cone and breaks into a stream of tiny droplets that are electrically charged.
The ions that are generated from the paper spray ionization technique can be coupled to a mass spectrometer for analysis. Such a precision instrument can reveal the molecular composition of the sample. Paper spray ionization technique is advantageous for producing ions from macromolecules because it creates very little fragmentation so its great for the analysis of proteins for instance. Paper spray ionization is a soft ionization technique and it is a variant of electrospray ionization.
The sample ions can also be deposited on a film. Once ions are generated by the paper spray ionization process, sample ions can be guided and deposited by manipulating the electric field potential. These dispersed samples can then be inspected under a microscope and analyzed in details. You can conduct many interesting experiments with this simple setup.
What is needed to generate ions using the paper spray ionization technique?
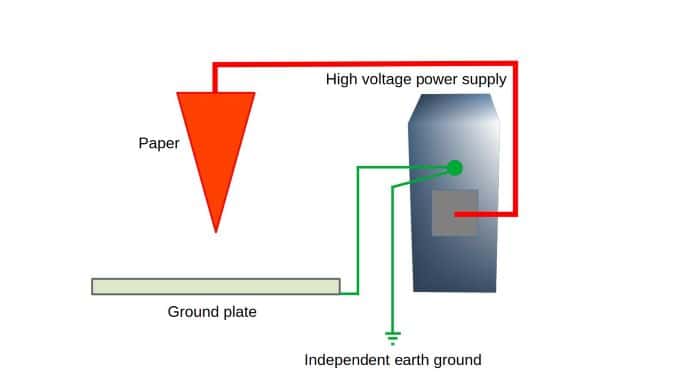
To do the paper spray ionization experiment, an adjustable high voltage power supply is needed. In this demonstration, we are using a benchtop high voltage power supply that can be adjusted up to positive 10 kilovolts. The other thing needed is a piece of paper that is connected to the high voltage power supply and held in a way that it is electrically floating above the metal plate that is grounded. As you can see, this setup is simple.
- Adjustable high voltage power supply
- Ethanol or any easily accessible alcohol to use as solvent
- Printer paper, paper towel or any common household paper
- Ground plate
Experiment results
The only special item used to conduct the experiment is the high voltage power supply. The rest of the things needed are common household items. The economical advantage of the paper spray ionization technique becomes apparent when dealing with high volume samples. when analyzing sensitive samples, it is very important to prevent cross-contamination. This is easy to accomplish with the paper spray ionization technique since the things needed to prepare and deliver the sample solution are consumable parts that are common and inexpensive. Experimental results are presented below.
Printer paper
Let’s start off with the printer paper for the first experiment. For the solvent, we are using 70% ethanol and we are applying positive 5 kilovolts. In cases where too much solvent is added to the printer paper, you get these instances of big droplets getting sprayed. After the excess solvent is drained then you get these really nice spraying cones that form at the tip. It creates very fine mist that are often hard to see and even more difficult to record but an intense light shining at certain angle helps a lot.
Is it possible to generate multiple spraying cones simultaneously? Yes, as you can see, multiple tips are cut into one continuous sheet of printer paper. When solvent solution comes in contact with the printer paper, wetting takes place quickly, spreading over the surface and absorbing into the porous paper structure. So the solvent that is added spreads across the sheet and to all the tips. When high voltage is applied, multiple spraying cones are formed simultaneously. Eventually, the solvent runs out and the paper spraying stops. This is a great way to increase the throughput of ion generation. As you can see, the mark on the aluminum ground plate is interesting since the pattern doesn’t overlap. There is a small gap and this is likely due to electrostatic repulsion of the charged droplets.
Overall, commercial printer paper works well for paper spraying.




Paper towel
The second case we will look at is the household paper towel, which also works well. Paper towels are selected and modified for maximum sorption of water into tissue material. We are conducting the paper spraying experiment with ethanol and high voltage power supply set at 5 kilo volts here as well.
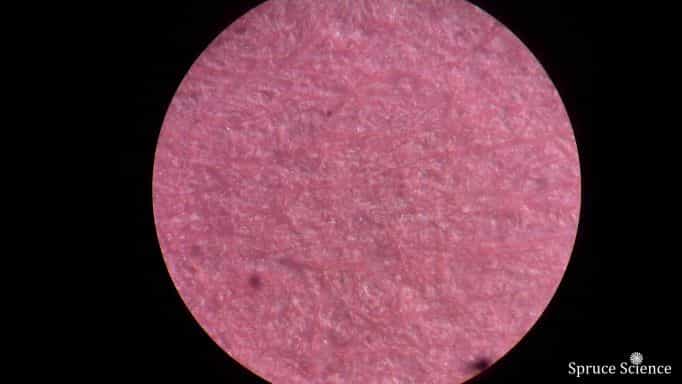
You can inspect the spraying performance by adding red ink. When 5 kilo volts is applied to the paper towel that holds the red ink solution, you will see red ink slowly accumulate on the piece of paper that sits on the ground plate. The paper sprayed sample can be inspected visually under a microscope. As you can see from the miroscope image, the red ink is very fine and evenly distributed.
If you change the solvent from ethanol to sake then voltage threshold for electrospraying goes higher. With Sake used as the solvent, you don’t see any spraying actions at 5 kilo volts. When high voltage output is adjusted upward to 7.6 kV then the spray cone forms and electrospray ionization takes place. Voltage shifted higher because sake is stickier since it contains many other ingredients.





WypAll
The third one we will test is the WypAll cleaning cloths from Kimberly-Clark. These cloths does not contain adhesives or binders and they don’t leave residues behind. The fabric in these cloths are pulp and polypropylene. It maybe great for cleaning but for paper spraying, the performance is mediocre. Under certain conditions, WypAll works and it does the spraying but sample delivery to the tip isn’t consistent so it oscillate between big droplets and fine mist. As shown here with the red ink, the spraying is inconsistent and splatters all over the collecting paper.




pH indicator strip
Let’s try using the pH indicator strip. As you can see, the pH indicator strip does spray very fine mist. However, it form multiple jets near the tip. It’s possible there are more than one obvious sharp point and more than one intense electric field that’s contributing to the formation of multiple jets from one tip.


Q-tip
Let’s deviate more from the paper theme and try Q-tip since they are also common household items. Unfortunately, Q tip doesn’t appear to be a good fit for electrospray ionization. When high voltage is applied, the fibers charge up to the same polarity and repel each other so you can see the in dividual fiber movements but there is no spraying and cone formation.


Cleanroom swab
This is a cleanroom swab with a foam head. Because of the foam material, it holds and release the solvent in a consistent manner so as you can see that it performs much better.


Metal wire tube brush
If we are trying to create multiple spraying source then what if we use something with a lot of sharp points like the metal wire tube brush that’s shown here? The cloth on the top is to help distribute and deliver the solvent to the wire and does not play a direct role here. It is difficult to get more than one cone forming and spraying at the same time. The solvent tends to form as big droplets on the wire and the electrostatic force pulls them towards the ground plate. When it does form a nice cone, it is unstable.
Here in this frozen frame, you can see the big droplet breaking up into smaller droplets midway and the fine mists around it.


Paint brush
Lastly, you can see the paint brush here but it doesn’t work that well. The spraying duration is short and the solvent doesn’t get released in slow consistent manner. It also doesn’t create multiple spraying cones although it has many sharp tips.


Conclusion
In conclusion, paper spray ionization setup is simple and you can potentially carry out many interesting experiments economically. You need an adjustable high voltage power supply but the rest are common materials.
We had fun running the benchtop experiments and it’s a pleasure sharing the results with you so I hope you enjoyed watching them.
Paper spray ionization technique video demonstration





